z-Pinch
I started thinking seriously about z-Pinches
after receiving an e-mail with some suggestions about my
earlier attempts at diamond synthesis. Tom Wahl asked if I thought
that crushing a hollow sphere of graphite with a high current pulse through
a coil (as in a can crusher) might lead to diamond formation. A few
calculations convinced me that this would not generate sufficient pressure
to convert graphite to diamond, but it got me thinking about how much pressure
could be generated via a z-pinch and whether or not this applied to the
process described in the Pike-Biegunsky
Patent.
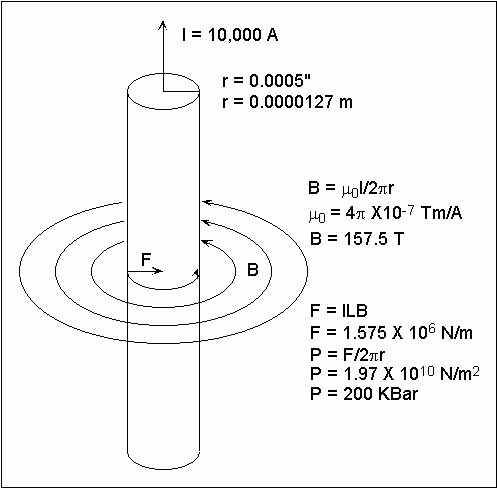
This drawing shows my analysis of the compressional force acting on
a 0.001" graphite fiber carrying a current of 10,000 Amperes. The
formulas can be found on the web at HyperPhysics.
I have made only one assumption, namely that the current is confined primarily
to the surface of the fiber. If we are using a current pulse source
such as a capacitor with a fast rise time, this should be true due to the
"skin effect". My calculations come up with a pressure of 200,000
atmospheres at the surface of the fiber. Is this sufficient to convert
graphite into diamond?
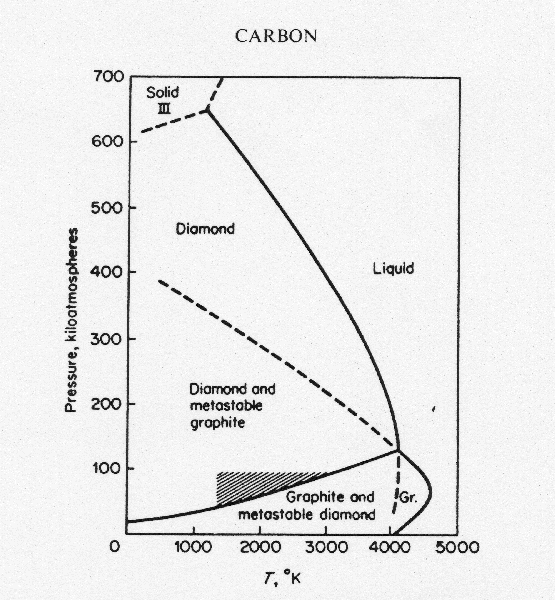
This is the phase diagram for carbon (taken from
"Inorganic Chemistry" by Cotton and Wilkinson). At one atmosphere
of pressure, graphite is the only thermodynamically stable form of solid
carbon. So why does diamond exist? There is also the issue
of kinetic stability. At reasonable temperatures the rate at which
the conversion takes place is essentially zero. However, at higher
temperatures, the rate increases. Heating a diamond to 4000º
Kelvin at one atmosphere of pressure (of an inert gas, like argon) will
convert it into graphite more or less instantly. Likewise, although
diamond is the only thermodynamically stable form of solid carbon at 200,000
atmospheres of pressure, the rate of conversion is slow below about 3000º
K. So, will a 10,000 Amp current heat a 0.001" graphite fiber to
at least 3000º K? Graphite has a resistivity of 1375.0 microohm-cm
at 20º C. If we have a one cm length of fiber, it will have
a resistance of (0.001375 W-cm)(1 cm)/p(0.00127cm)2
= 271 W. The rate of power dissipation
will be (10,000 Amps)2(271 W) = 27.1
X 109 Joules/second. At 3000º K graphite has a heat
capacity of 2.5 Joules/gramºK. Our hypothetical fiber contains
1.3 X 10-5 grams of graphite. To raise its temperature
from 295º K to 3000º K will require (2.5 J/gºK)(1.3 X 10-5
g)(2705º K) = 0.09 Joules of energy. At a power level of 27
GW, this will take only 3.3 X 10-12 seconds. No problem.
I am convinced that this method of diamond synthesis works because of the
z-pinch phenomenon.
Home


